NAFDAC halts registration of artemether/lumefantrine dry powder suspensions
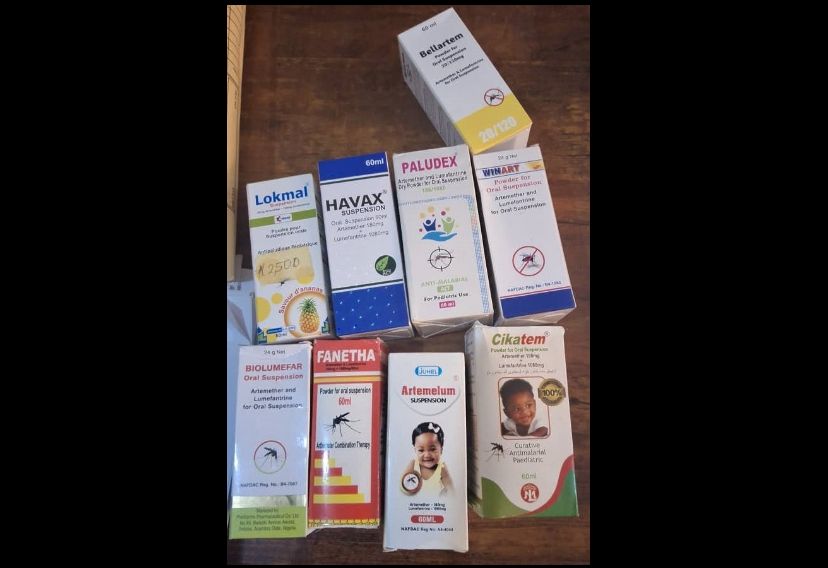
NAFDAC has reaffirmed its directive discontinuing registration of artemether/lumefantrine dry powder for oral suspension, warning the formulation becomes unstable after mixing and loses efficacy. The agency's directive, first issued in March 2025, applies to all locally manufactured and imported multi-dose suspensions. Stability studies confirmed reconstituted suspensions lose effectiveness, risking treatment failure and complications. Manufacturers must switch to safer single-dose alternatives like sachets. Report suspected sales of discontinued products via NAFDAC's toll-free line 0800-162-3322.